ABOUT US
Where Innovation Meets Better Health
At Klim-Loc, we’re redefining the future of healthcare with thoughtful, needle-free pharmaceutical technologies. Our work pushes boundaries to deliver solutions that enhance safety, comfort, and care, for providers and patients alike.
.png)
Industry report
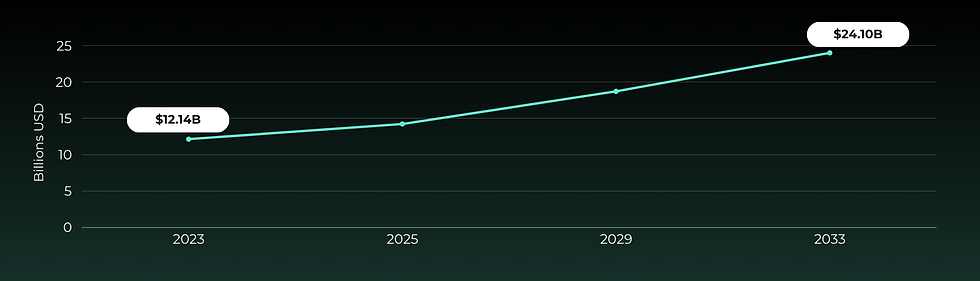
Global Pharmaceutical Glass Vial & Cap Market
The global pharmaceutical vial cap market will grow at a CAGR of 7.1%, doubling from $12.14 billion in 2023 to $24.1 billion in 2033. We expect to capture significant market share with our innovating cap alternative.

key partnerships
Market Validation
Klim-Loc demonstrates strong market validation through letters of support, DoD consortia, and key partnerships. These endorsements reflect clear demand and a trusted path to adoption across military and civilian healthcare systems.
Endorsements and early adoption interest from leading industry experts further validates the Klim-Loc product viability.
Jake Sangster Intermountain Ventures, Karl Cazzini, Ph.D. Director Engineering, Innovation & Exploratory Baxter, Connor Feeney M&A Analyst Medline, A. Robb McClean, MD NMU School of Medicine, Tom Feller & Kelly Thompson CareFlight of the Rockies.


Development Plan with Our CRO Partner
Comprehensive Medical Device Design & Development Services
Our partner offers a full suite of services, from concept development to manufacturing, to bring medical devices to market efficiently & effectively.
Regulatory Expertise & Compliance
Their team of experts navigates the complex regulatory landscape, ensuring devices meet all relevant standards & guidelines.
Cutting-Edge Prototyping & Testing Capabilities
Our partner's state-of-the-art facilities allow for rapid prototyping, rigorous testing, & iterative design refinement.
Successful Track Record of Commercialized Products
They have a proven history of helping clients successfully bring innovative medical devices to market, with a portfolio of commercially available products.
Their deep regulatory knowledge and integrated design-to-manufacturing services position Klim-Loc for efficient validation, FDA submission, and scalable production.
Go-to-Market Strategy
Steps I & II
Military & Government (Step I)
Collaborate with the DoD and personnel in austere environments, integrating vial caps into medical countermeasure and vaccination programs with BARDA and other consortia.
Hospital & Healthcare Facilities (Step I)
Agnostic to site of care: hospitals, operating rooms, emergency rooms, ICU’s, ambulatory surgery centers, first responders, outpatient offices, home health.
Pharmaceutical Companies (Step I)
Partner with pharmaceutical companies to integrate our needleless vial cap system with medications and existing fill-and-finish facilities.
Veterinary Care & Services (Step II)
Veterinary clinics and animal hospitals as well as large animal field based veterinary practices.
Klim-Loc’s revolutionary technology streamlines medication and IV fluid delivery, improving safety, speed, and efficiency across all healthcare settings. Universally applicable and highly scalable, Klim-Loc is positioned to capture significant market share through strategic partnerships and licensing, becoming the global standard in medicine delivery.
How do we drive revenue?
B2B Model
Regulatory Readiness
Advance toward FDA clearance and Type III DMF submission, positioning Klim-Loc for scalable commercialization and rapid partner adoption.
B2B Sales
mplement targeted marketing campaign towards pharmaceutical (fill + finish), hospitals, and DoD.
Strategic Partnerships
Expand industry relationships to include CMO’s, wholesale distributors, and aligned partners.
Diversify Revenue Streams
Explore new revenue opportunities: exclusivity contracts, and licensing.
By focusing on these key strategies, we will create a sustainable and profitable business model that delivers long-term growth and financial success. Established interest & potential early adopters: Baxter International, B.Braun, Gerresheimer, DoD.
_edited.jpg)
Competitive Landscape
Key player in the vial rubber stopper market
These top 4 companies account for 70% of theglobal pharmaceutical vial rubber stopper market.

We are disrupting this entire multi-billion dollar market.
_edited.png)
Our innovative and highly differentiated vial cap will be comparably priced to products currently on the market providing Klim-Loc with a clear competitive advantage.

Future Product Expansion
A pipeline of revolutionary, sensor-integrated products designed to work synergistically, expanding impact, efficiency, and value, across healthcare applications.
Spikeless & Needleless IV
Fluid Bag & Infusion Set
Eliminates the need to 'spike' IV fluid bags by replacing the spike port with a patented threaded port system for seamless infusion set coupling. IV bag threaded accessory port provides a needleless site to extract IV fluid and to inject medications with a Luer-Lock syringe.
Innovative, efficient, and safe.
patent filed & working prototype
Sensor Technology
Real-time data collection of pharmaceutical and fluid administration.
Auto populates data into EMR: time stamp, personnel administering, medication administered, volume & dose, lot#, expiration date, usage rate, and more.
Relieves charting, administrative, logistical, and supply chain burdens.
Value Proposition
How we drive broad pharmaceutical market adoption
Klim-Loc’s innovative design preserves existing value-chain structures, minimizes switching costs for pharmaceutical manufacturers, and simplifies regulatory pathways for faster market adoption.
Seamless Workflow Integration
Proven Luer based components seamlessly integrate into well established and ubiquitous global pharmaceutical work-flows with no training required.
Integration with Existing Fill-&-Finish Lines
Innovative design allows the needleless vial cap to adapt to existing fill-&-finish lines and aluminum crimp cap container closure system process.
Needleless Vial Cap Design
Fits with numerous existing pharmaceutical vial neck and volume sizes as well as fits within existing pharmaceutical industry dispensing and storage bins.
Utilization of Existing Approved Materials
Manufactured from ISO-compliant and regulatory approved polycarbonate, rubber, & elastomeric materials.